InsightsElectronic Assemblies: Risk Factor Electrochemical Migration
An Overview of Basics and Mechanisms of Electrochemical Migration on PCBs
Failure CauseElectrochemical Migration
Electrochemical Migration (ECM) in electronics compromises the reliability of assemblies, often due to climate-related failures. It results from the movement of metal ions due to electric fields and moisture. This migration can lead to temporary malfunctions and permanent short circuits, and in extreme cases, it can even cause overheating and fires. Understanding the conditions of occurrence and the mechanism is crucial for implementing preventive measures
understanding electrochemical migration ON PCBsThe Conditions of Its Occurrence
Electrochemical Migration is favored by moisture, enabling corrosion. It occurs through moisture films on surfaces or dew formation, with the materials used and possible contaminants playing a significant role.
The critical humidity for moisture films depends on the surface energy and polarity, i.e., the material, especially the solder mask. This critical threshold can be reached even at humidity levels below the dew point, as minimal film thicknesses are enough to cause corrosion.
Dew formation is induced by temperature changes and focuses on the areas with highest probability of condensation such as metallization or contaminants. Solder residues like organic acids or halide salts locally lower the dew point down to 60% relative humidity.
The material used is a crucial factor. Moisture films form on metals or metal oxide surfaces at a critical humidity of 60%-70% RH, while on aluminum oxide ceramics, they only form at over 90% RH. Additionally, the metallization or solder material must have an active region in alkaline electrolytes for migration to occur. Certain metals tend to migrate, while others remain unaffected under similar conditions. The susceptibility of each element must be assessed.
Contaminants on assemblies, such as flux residues or dust, serve as condensation nuclei and promote dew formation. They can retain moisture on surfaces and hinder the redrying of polymers, for example.
Mechanism of electronical migration ON PCBsThe Process of ECM Occurs in Three Key Steps:
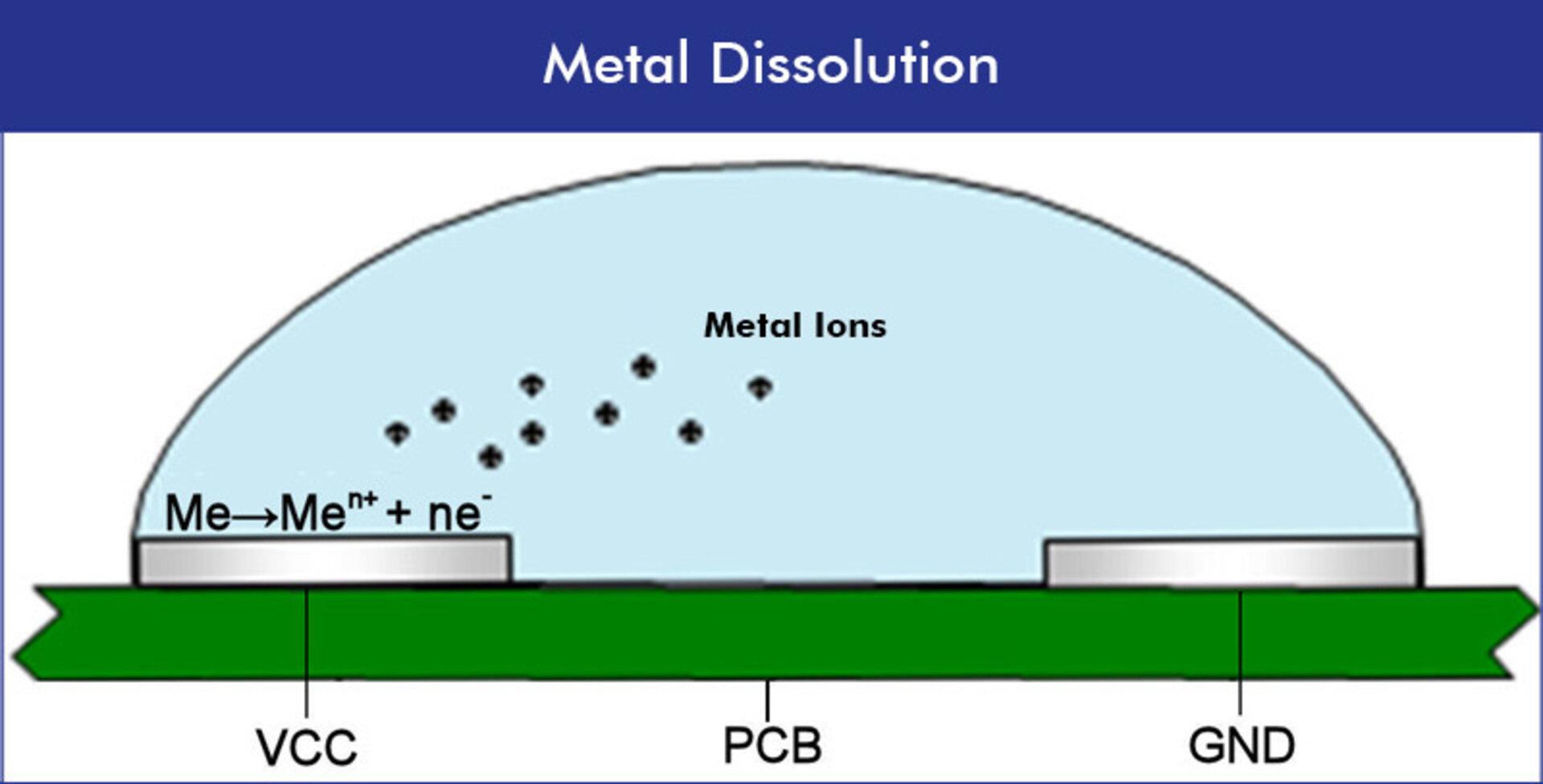
01 | Anodic Metal Dissolution
Moisture films on assembly surfaces reduce surface resistance and thus insulation capability. Above a critical film thickness, electrolysis occurs, leading to local alkalinization at the anode, so that metal ions such as silver, copper, tin, and lead get electrochemically activated. The dissolution of the anode surface generates metal complex concentrations that spread along a concentration gradient.
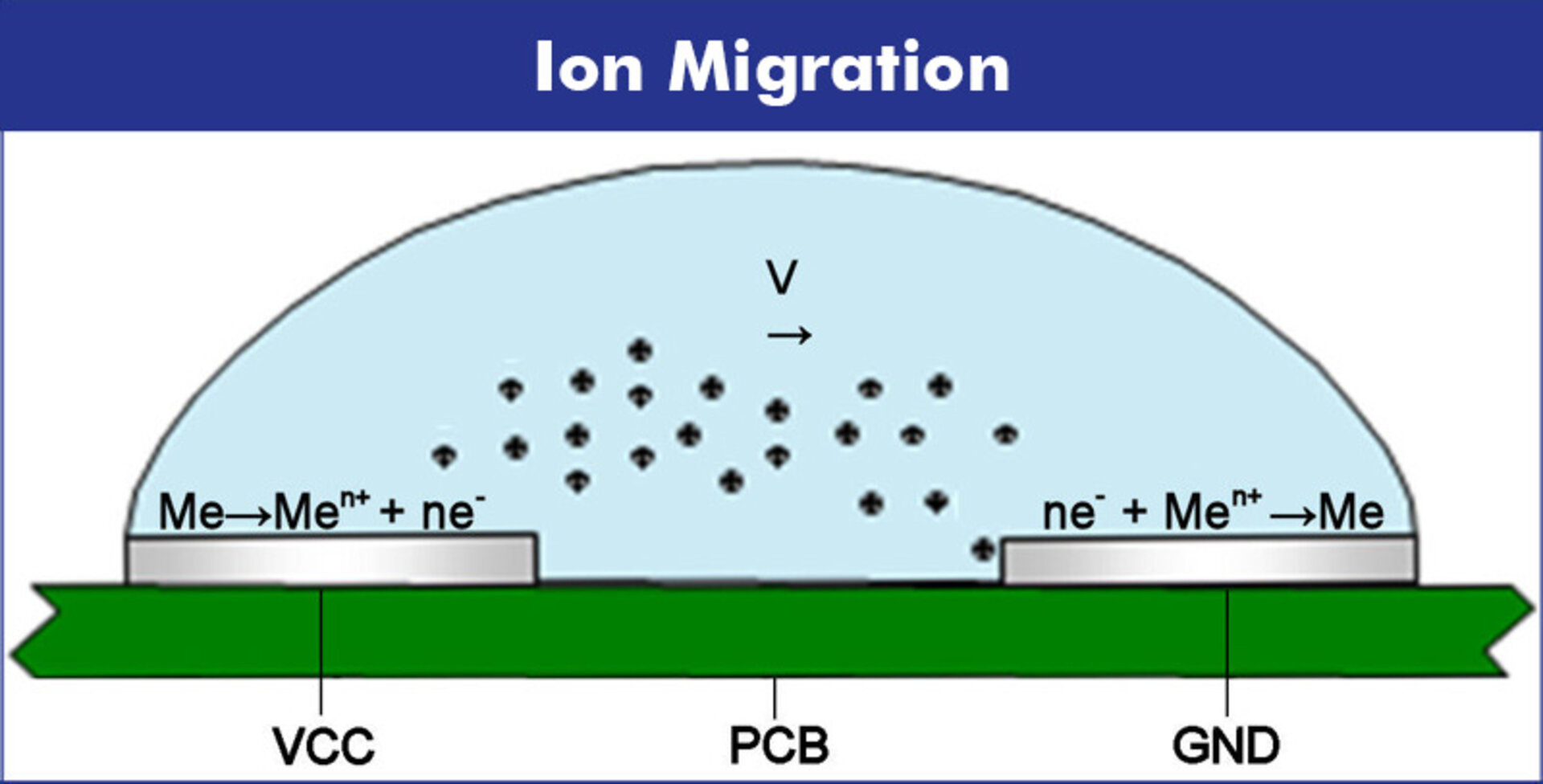
02 | Migration of Metal Ions
Ion movement is controlled by the electric field, respectively the potential gradient and the concentration gradient. The potential gradient depends on the operating voltage and conductor distance, while the concentration gradient is influenced by the dissolution and diffusion speed of solvated metal ions. The ratio of these two gradients determines whether the ions diffuse along the concentration gradient or even migrate against the electric field from mass to contact.
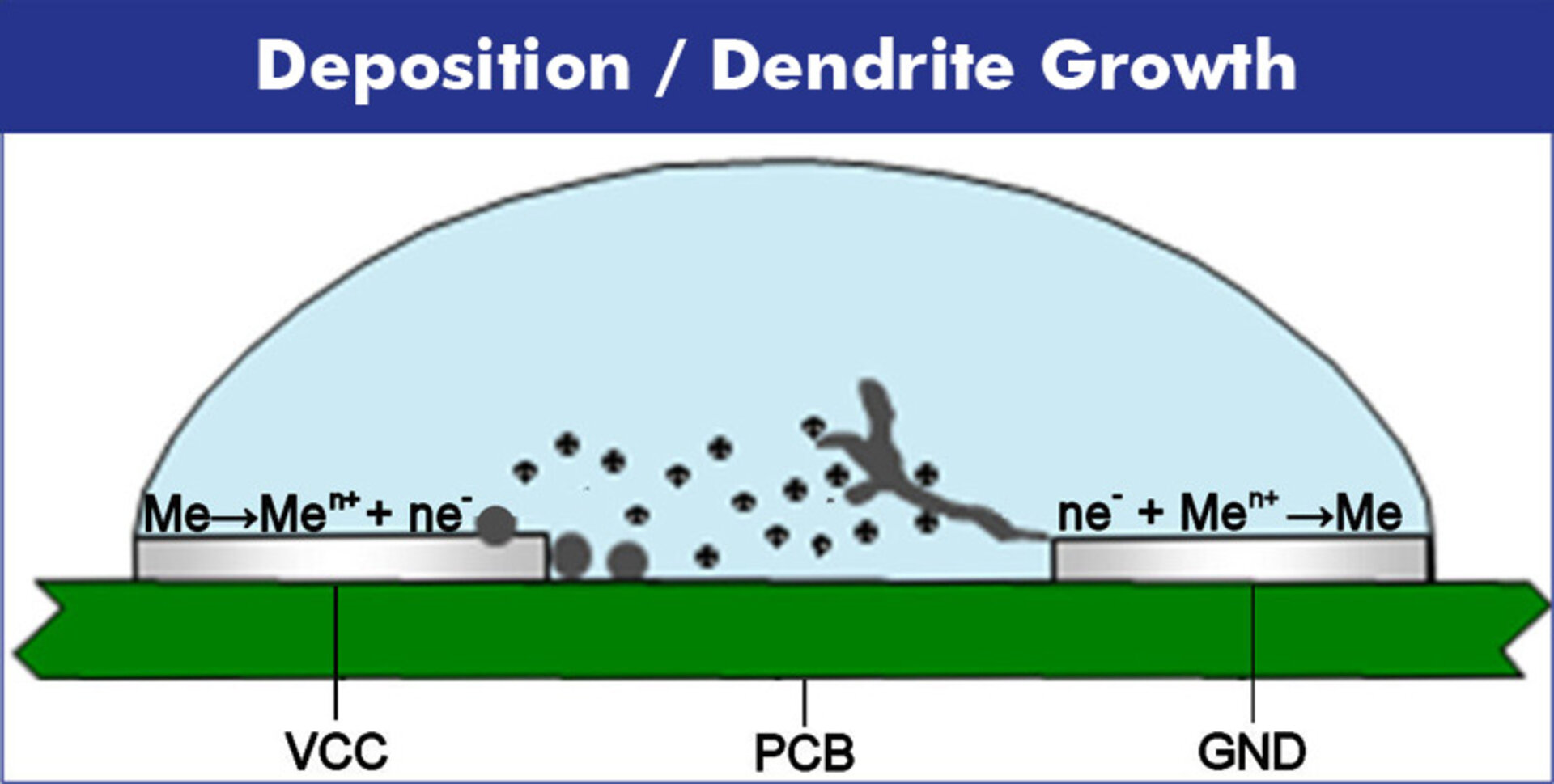
03 | Depostion of Metal Ions
Bridge formation occurs either through galvanic deposition at the cathode or by the precipitation of hydroxides, oxyhydrates, or complexes as salts at the anode. The structure of the bridges varies depending on ion concentration and field strength and can take dendritic structures or smoother growth forms.
Elektrochemical migrationDifferentation from Voltage Breakdown and Analyzing Effects
Distinguishing ECM from Other Damage Patterns
To address failures specifically, it is important to distinguish Electrochemical Migration from other failures such as voltage breakdowns or graphitizations on components. Voltage breakdowns are mainly caused by pores in the solder resist, reducing insulation capability. Graphitizations on components occur when the insulating effect of organic coatings is too small. The cause here also lies in porosity, which must be addressed by optimizing the coating.

Consquences of Electochemical Migration on PCB
In case of failures or disturbances, it is often difficult or only with great effort possible to prove that Electrochemical Migration is the cause.
During short dew times, small dendrites are formed, which cannot carry high currents and burn immediately. This leads to user dissatisfaction and potential high follow-up costs. The proof often requires elaborate investigations, which are logistically challenging. As a result, ECM failures in the field often remain unidentified and are mixed with other issues such as software errors or leakage current.
However, if persistent dendrites form, temperatures of several hundred degrees Celsius can be quickly reached. Without adequate protection, these can lead to fires and destroy the circuit. Afterwards, it is often speculative whether the cause was Electrochemical Migration or electrical breakdowns.

ContactWe are Pleased to Assist You
Would you like to know how to prevent the occurrence of ECM on your assembly? Are you experiencing (field) failures and need support in analysis and remedy?

zestron insightsElectrochemical Migration: Tracking Power Module Failures
Read about how our R&S specialists addressed quality issues with dishwasher assemblies. Discover more about our effective solution approaches in our Case Study.
Whitepaper-Collection Electrochemical Migration
Our free articles/whitepapers on Electrochemical Migration provide answers to questions about the origin and behavior of this mechanism. Learn, among other things, how the right protective measures can effectively prevent or reduce Electrochemical Migration (ECM).